Sanofi’s New CEO Taking the Helm: What Will Be the Growth Strategy?
Merck KGaA Chair and CEO Belén Garijo is coming on board as Sanofi’s new CEO next week, succeeding former CEO Paul Hudson, who stepped down in February. The company reported its first quarter results this week? What commercial and pipeline products will drive growth?
By Patricia Van Arnum, Editorial Director, DCAT, pvanarnum@dcat.org

Changing of the guard at Sanofi
Belén Garijo, now Chair of the Executive Board and CEO of Merck KGaA, takes the helm next week (April 29, 2026) as CEO of Sanofi. She faces the task of driving revenue growth through increased product diversification and R&D productivity as the company remains heavily reliant on its top-selling product, Dupixent (dupilumab), a drug for treating multiple inflammatory diseases. She succeeds Sanofi’s former CEO, Paul Hudson, who stepped down from the company in February (February 2026) with Olivier Charmeil, Executive Vice President, General Medicines, serving as Interim CEO during the transition.
In his role as interim CEO, Charmeil outlined the company’s first-quarter 2026 results, which were released this week (April 23, 2026). The company reported favorable results with net sales of EUR 10.5 billion ($12.3 billion), an increase of 13.6% year-over-year at constant exchange rates (CER) (6.2% at actual exchange rates). The key revenue driver was Dupixent, which posted first-quarter 2026 net sales of EUR 4.2 billion ($4.9 billion), a 30.8% gain year-over-year.
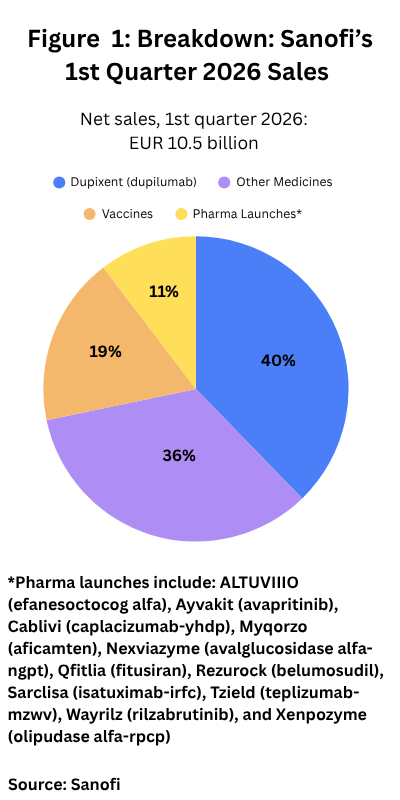
Duxipent accounted for 40% of Sanofi’s first-quarter 2026 revenues (see Figure 1) and is the company’s top-selling product with 2025 sales of EUR 15.7 billion ($18.4 billion), accounting for 36% of its 2025 full-year revenues of EUR 43.6 billion ($51.0 billion). Other top-selling products for the company in 2025 were its long-acting insulin products, Toujeo (insulin glargine), a next-generation version of the company’s Lantus (insulin glargine), which faces biosimilar competition. Toujeo posted 2025 sales of EUR 1.35 billion ($1.58 billion) and Lantus EUR 1.73 billion ($2.02 billion). Fabrazyme (agalsidase beta), an enzyme replacement therapy for treating Fabry disease, a rare lysosomal storage disease, and which faces biosimilar competition in certain markets, posted 2025 sales of EUR 1.02 billion ($1.19 billion).
Sanofi is banking on new/recent product launches to drive growth (see Table I at end of article). Among the best performers thus far are Beyfortus (nirsevimab-alip), ALTUVIIIO (antihemophilic factor (recombinant), Fc-VWF-XTEN fusion protein-ehtl, previously referred to as efanesoctocog alfa), and Nexviazyme/Nexviadyme (avalglucosidase alfa-ngpt), which posted blockbuster sales (defined as sales of $1 billion or more) or near blockbuster status in 2025.
Beyfortus, which Sanofi co-developed and co-commercialized with AstraZeneca, is a preventive antibody against respiratory syncytial virus lower respiratory tract disease in newborns and infants. It posted full-year 2025 sales of EUR 1.78 billion ($2.08 billion) and first-quarter 2026 sales of EUR 284 million ($332 million). ALTUVIIIO is a factor VIII replacement therapy for routine prophylaxis and on-demand treatment to control bleeding episodes for adults and children with hemophilia A. It had 2025 sales of EUR 1.16 billion ($1.35 billion) and first-quarter sales of EUR 325 million ($380 million). Nexviazyme/Nexviadyme, an enzyme-replacement therapy for treating Pompe disease, a rare, inherited metabolic disorder, had 2025 sales of EUR 790 million ($924 million) and first-quarter 2026 of EUR 208 million ($243 million).
Whether these and other upcoming product launches (see Table 1 at end of the article) will materialize is a key question for the company. New recent/product launches accounted for only 11% of the company’s first-quarter 2026 sales (see Figure 1), which included ALTUVIIIO and Nexviazyme and some others which have not yet had seen strong sales since initial approval.
On the potential upside are Ayvakit (avapritinib), which Sanofi gained via its $9.5-billion acquisition of Blueprint Medicines in 2025, in a move to boost Sanofi’s rare-disease drug portfolio. The drug is approved for treating adults with indolent or advanced systemic mastocytosis, a rare blood cancer, and specific gastrointestinal stromal tumors.
Driving product innovation is strategic priority of the company and in naming Garijo as CEO. “Belén Garijo will bring an increased rigor to the implementation of Sanofi’s strategy and accelerate the preparation of the Group’s future. Her priority will be to strengthen the productivity, governance, and innovation capacity of Research & Development,” said the company in the February 12, 2026 release.
Table 1: Tracking Innovation: Sanofi’s Recent Product Launches
| Brand name (active pharmaceutical ingredient); Modality | 2025 Net Sales | 1st Quarter 2026 Net Sales | Initial US approval |
| Beyfortus (nirsevimab-alip)* Biologic | EUR 1.781 Bn ($2.08 Bn) | EUR 284 M ($332 M) | 2023 |
| ALTUVIIIO* (efanesoctocog alfa) | EUR 1.160 Bn ($1.35 Bn) | EUR 325 M ($380 M) | 2023 |
| Nexviazyme/Nexviadyme (avalglucosidase alfa-ngpt) | EUR 790 M ($924 M) | EUR 208 M ($243 M) | 2021 |
| Sarclisa (isatuximab-irfc) Biologic | EUR 588 M ($688 M) | EUR 167 M ($195 M) | 2020 |
| Rezurock (belumosud)* Small molecule | EUR 490 M ($573 | EUR 133 M ($155 M) | 2021 |
| Ayvakit (avapritinib)* Small molecule | EUR 305* M ($357 | EUR 177 M ($207 M) | 2020 |
| Cablivi (caplacizumab-yhdp)* Biologic | EUR 271 M ($371 | EUR 68 M ($79 M) | 2019 |
| Xenpozyme (olipudase alfa-rpcp) Biologic | EUR 228 M ($267 | EUR 63 M ($74 M) | 2022 |
| Tzield / Teizeild* (teplizumab-mzwv) Biologic | EUR 63 M ($74 | EUR 14 M ($16 M) | 2022 |
| Nuvaxovid (mRNA adjuvanted COVID-19 vaccine) | EUR 29 M ($34 | EUR 16 M ($19 M) | 2025 |
| Qfitlia (fitusiran) Small molecule | EUR 9 M ($10.5 | EUR 5 M ($5.8 M) | 2025 |
| Wayrilz (rilzabrutinib) | EUR 7 M ($8.2 | EUR 10 M ($11.7 M) | 2025 |
| Heplisav-B (Hepatitis B vaccine (recombinant, adjuvanted) | N/A | EUR 46 M ($54 M) | 2017 |
| Myqorzo (aficamten) | N/A* | EUR 1 M ($1.2 M) | 2025 |
Bn is billions; M is millions; N/A is not applicable.
Beyfortus. AstraZeneca and Sanofi co-develop and commercialize nirsevimab in an agreement formed in 2017.
Rezurock. Sanofi gained Rezurock from its acquisition of Kadmon Holdings in 2021.
Ayvakit. Sanofi gained Ayvakit via its acquisition of Blueprint Medicines in June 2025. Net sales are on a proforma basis; full-year sales were $725 million.
Cablivi. Sanofi gained Cablivi from its acquisition of Ablynx in 2018.
Tzield / Teizeild. Sanofi gained Tzield / Teizeild through acquisition of Provention Bio in 2023.
Nuvaxovid. Novavax transferred US marketing authorization for Nuvaxovid to Sanofi in November 2025. The vaccine received emergency use authorization by US Food and Drug Administration in 2022 and full FDA approval in 2025.
Heplisav-B. Sanofi acquired Heplisav-B through its of Dynavax Technologies in late 2025.
Myqorzo. Cytokinetics is responsible for the commercialization of Myqorzo (aficamten) in the US and Europe. Sanofi holds excluive right in Greater China via an agreement with Corxel Pharmaceuticals, who had acquired them from Cytokinetics.
Source: Sanofi






