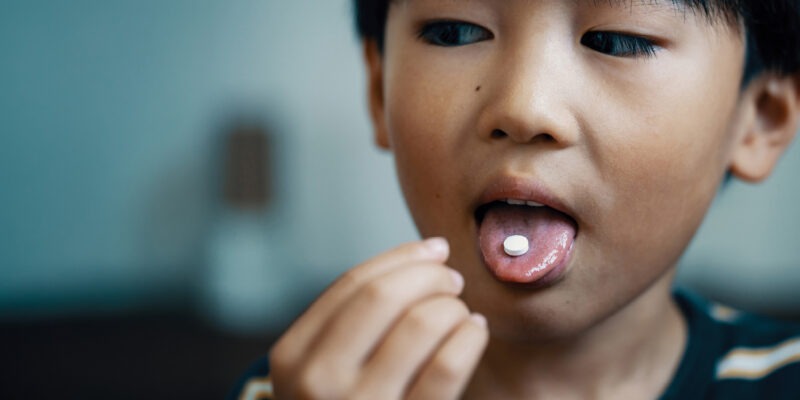
A new way of tackling bitterness and elevating drug palatability: ModulaSENSE® Bitter debuts at DCAT Week
By Carlos Almasqué, Global Marketing Director Pharma Excipients, dsm-firmenich and Dr. Pierre-Louis Bardonnet, Global Technical Business Development Manager Pharma, dsm-firmenich
As pharmaceutical drug formulations grow more complex, the challenge of creating palatable medications continues to stand between innovation and patient adherence. ModulaSENSE® Bitter—which was introduced at DCAT Week—represents a new, science‑driven way forward to address this issue. This breakthrough technology from dsm-firmenich tackles the bitterness of active pharmaceutical ingredients (APIs) at the receptor level, helping formulators improve drug palatability and support patient compliance. It is a clear demonstration of the company’s expanding portfolio of pharma‑grade excipients and comprehensive formulation capabilities.
The measurable impact of taste on compliance
The palatability of medications plays a critical role in patient adherence, with consequences that go far beyond unpleasant taste. Consumers themselves recognize the issue; around 40 percent agree that taste influences their choice when purchasing over-the-counter products (1). In addition, up to 64 percent of patients are at risk of discontinuing treatment as a result of poor taste (2). This issue is also acknowledged by regulatory bodies around the world—including the European Medicines Agency (EMA) and the U.S. Food and Drug Administration (FDA)—which emphasize the importance of taste and palatability (3, 4). Improving the palatability of medicines is therefore a key challenge for the pharmaceutical industry, particularly in oral dosage forms addressing pediatric and geriatric populations.
“Palatability is defined as the quality of a drug product that makes it pleasant or acceptable in terms of taste, after-taste, smell, and texture, and it is a critical factor in determining patient acceptance of oral dosage forms (5).”
The biology of bitterness
Humans are biologically adapted to repel unpleasant tastes, particularly bitterness, which is thought to serve as a warning mechanism for toxic substances found in nature. Bitterness is detected by a family of 25 taste receptors known as bitter taste-sensing type 2 receptors (TAS2Rs or T2Rs), and it is a common problem in the pharmaceutical industry, with over 60 percent of APIs being intrinsically bitter (6, 7). This can lead to non-compliance with treatment regimens and suboptimal treatment outcomes and is further complicated by the fact that perception of bitterness varies from one individual to another (8). Unfortunately, there is no one-size-fits-all approach to bitterness masking as formulation constraints must be carefully managed to maintain therapeutic efficacy, making the development of truly patient-friendly dosage forms a significant challenge.
ModulaSENSE® Bitter: Targeting bitterness at the receptor level
ModulaSENSE® Bitter takes a novel approach to tackling bitterness in drug formulations, harnessing proprietary, receptor-based discovery methods and sensory validation to elevate drug palatability and, ultimately, help to improve patient adherence. Instead of relying on generic masking, ModulaSENSE® Bitter combines blockers, maskers, and sweeteners with evidence-based insights to create targeted, formulation-specific bitterness masking solutions. The technology has been developed through a multi-step, scientifically-driven workflow and is applicable across a wide range of APIs and oral dosage forms. The result is improved palatability through targeted bitterness reduction, improving patient acceptance across demographics, including pediatric and elderly populations. Crucially, ModulaSENSE® Bitter is supported by comprehensive technical documentation and a regulatory package as well as sensory evaluation. This approach helps to streamline formulation development while meeting patient and healthcare professionals’ expectations for better tasting therapies.
Beyond taste: an integrated pharma toolkit
ModulaSENSE® Bitter builds on a solid foundation of pharma-grade solutions at dsm-firmenich, including a specialized flavors and taste modulation portfolio that has been carefully designed for pharmaceutical drug formulations. This portfolio is rich and varied, from flavors and extracts to aromatic compounds—encompassing core notes such as citrus and mint, plus more intricate nuances—maskers, blockers, and sensates. This portfolio has been crafted by a dedicated pharma team to precisely meet customers’ technical and regulatory requirements, streamlining drug development, and enhancing brands with quality formulations.
The drug formulation process is becoming increasingly complex due to the rise of novel APIs and advanced drug delivery systems. While flavors are part of the solution, there is a need for more innovative approaches to drug development, where excipients play a multifaceted role—from contributing to drug stability and solubility to improving bioavailability. Alongside its taste solutions, dsm-firmenich offers a high-quality specialty excipients and ingredients portfolio—including GMP-compliant vitamins and vitamin derivatives—which can help to overcome even the most difficult formulation challenges.
Vitamins as functional excipients
Vitamins serve as versatile excipients in pharmaceutical drug formulations. They can play a critical role in improving the profile of small molecules by enhancing API stability and overall formulation performance. Additionally, vitamins and vitamin derivatives act as stabilizers, adjuvants, and nutrients in biologic formulations, supporting advanced therapies and biopharmaceutical cell cultures. Their multifunctional properties make them essential components in overcoming complex formulation challenges, of which two examples are discussed below.
Nitrosamines have been flagged as potential carcinogens and were discovered in common drugs—such as ranitidine, nizatidine, and metformin—in 2018. This led to increased scrutiny by regulators, which now mandate the adoption of procedures to evaluate, measure, and mitigate nitrosamines in drug products. The use of nitrite scavengers as functional excipients in drug formulations could be one way of inhibiting the formation of nitrosamines. However, not all nitrite scavengers are suitable for use in pharmaceutical formulations, and only a few are listed on the FDA’s inactive ingredient database (9). Ascorbic acid (vitamin C) and alpha-tocopherol (vitamin E) are specifically mentioned by the US regulator as possible strategies to mitigate nitrosamine formation (10). These vitamins are therefore increasingly being explored and used as excipients in pharmaceutical products, combining well-established safety profiles with unique properties to overcome complex formulation challenges. Incorporating even low levels of ascorbic acid could significantly reduce the risk of nitrosamine formation while the quality, grade, and purity of any added ingredients should be considered carefully to ensure optimal efficacy and safety of the final formula.
Vitamins with antioxidant properties can also be incorporated into formulations to enhance stability or protect sensitive drugs from degradation caused by various mechanisms. For example, antioxidants are being explored as excipients in radiopharmaceuticals. These products face a unique set of logistical challenges—including difficult supply chains, high costs, and complex administration. But stability remains the most critical issue as products can degrade over time due to radiolysis, generating free radicals that compromise pharmaceutical purity, efficacy, and safety. Sodium ascorbate has been shown to be a highly effective free radical scavenger, neutralizing reactive species and protecting radiopharmaceuticals against degradation. This not only preserves the product’s integrity, but it also extends its shelf life (11, 12). The application of these well-known and characterized compounds as excipients is proving vital to support radiopharmaceutical innovation in emerging fields such as precision medicine. As this field continues to diversify—including the development of advanced therapy medicinal products—vitamin and vitamin derivatives will play an increasingly essential role in formulation development across many key applications.
A trusted partner across your formulation journey
Drug development is a lengthy and complex process, often spanning years of research, clinical testing, and regulatory review before reaching patients. This makes it essential to draw on the experience and knowledge of specialist partners along the way to accelerate approvals. dsm-firmenich offers more than just industry-leading innovation capabilities and cutting-edge solutions. Each product is backed by dedicated quality and regulatory teams, providing access to comprehensive documentation and regulatory support. This extends to major markets around the world; in-depth knowledge of the filing process in different regions helps customers to navigate the ever-evolving pharmaceutical market with confidence and peace of mind. dsm-firmenich partners with customers all along the development pathway, helping them to formulate with success and, ultimately, bring safe and effective drug products to patients worldwide.
References
- dsm-firmenich. Firmenich Taste Lounge Online Consumer Community, Total Sample n-200, September 2021. Published online 2021.
- Nordenmalm S, Kimland E, Ligas F, et al. Children’s views on taking medicines and participating in clinical trials. Arch Dis Child. 2019; 104(9):900-905. doi:10.1136/archdischild-2018-316511.
- U.S. Food and Drug Administration. (2023). Pediatric Drug Development Under the Pediatric Research Equity Act and the Best Pharmaceuticals for Children Act: Scientific Considerations. Available at: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/pediatric-drug-development-under-pediatric-research-equity-act-and-best-pharmaceuticals-children-act
- European Medicines Agency. (2013). Pharmaceutical development of medicines for paediatric use – scientific guideline. Available at: https://www.ema.europa.eu/en/pharmaceutical-development-medicines-paediatric-use-scientific-guideline.
- U.S. Food and Drug Administration. (2018). Use of Liquids and/or Soft Foods as Vehicles for Drug Administration: General Considerations for Selection and In Vitro Methods for Product Quality Assessments. Available at: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/use-liquids-andor-soft-foods-vehicles-drug-administration-general-considerations-selection-and-vitro.
- Dagan-Wiener A, Nissim I, Ben Abu N, et al. Bitter or not? BitterPredict, a tool for predicting taste from chemical structure. Sci Rep. 2017;7(1):12074. doi:10.1038/s41598-017-12359-7.
- Tisi DA, Worthington JH. Taste Masking Challenge of 155 Active Pharmaceutical Ingredients. Medical Research Archives. 2024;12(10). doi: https://doi.org/10.18103/mra.v12i10.5890.
- Wooding SP, Ramirez VA, Behrens M. Bitter taste receptors: Genes, evolution and health. Evolution, Medicine and Public Health. 2021;9(1):431-447. https://doi.org/10.1093/emph/eoab031
- U.S. Food and Drug Administration. (2026). Inactive Ingredients Database Download. Available at: https://www.fda.gov/drugs/drug-approvals-and-databases/inactive-ingredients-database-download.
- U.S. Food and Drug Administration. (2021). Updates on possible mitigation strategies to reduce the risk of nitrosamine drug substance-related impurities in drug products. Available at: https://www.fda.gov/drugs/drug-safety-and-availability/updates-possible-mitigation-strategies-reduce-risk-nitrosamine-drug-substance-related-impurities#2.
- Smith TA, Kirkpatrick DR, Smith S, et al. Radioprotective agents to prevent cellular damage due to ionizing radiation. Radioprotective agents to prevent cellular damage due to ionizing radiation. J Transl Med 15, 232 (2017). https://doi.org/10.1186/s12967-017-1338-x.
- Liu S, Ellars CE, Edwards DS. Ascorbic Acid: Useful as a Buffer Agent and Radiolytic Stabilizer for Metalloradiopharmaceuticals, Bioconjugate Chemistry. 2003;14(5): 1043-1802. doi: 10.1021/bc034109i.