Drug Pricing Reform: The Impact on Drug Development
The Congressional Budget Office (CBO) released a simulation model for analyzing the impact of US legislative drug pricing proposals on new drug development. The CBO projects that certain proposals, if adopted, may reduce the number of drugs entering the US market in the long term by as much as 59. Which policy proposals are most impactful?
Delving deeper into drug pricing proposals
One of the major legislative proposals on the table is a bill passed by the US House of Representatives in late 2019, the Elijah E. Cummings Lower Drug Costs Now Act (H.R. 3), but which has not yet been taken up in the US Senate. The key measures in the bill would require the US Department of Health and Human Services (HHS) to negotiate drug prices directly with manufacturers, subject to a cap based on international reference pricing. Discussions on the bill are ongoing and are expected to move forward as the US Congress considers other legislative proposals in the context of the federal budget process and overall as well as statements made by President Joe Biden last month (August 2021) calling on Congress to address the cost of prescription drugs. Although not providing specific details, the President indicated his support for measures to allow the federal government to negotiate drug prices under Medicare (the US healthcare program for people over the age of 65) and to firm a cap on the amount that Medicare beneficiaries have to pay out-of-pocket for drugs each year.
The proposed legislation (H.R. 3), analyzed by the CBO, would establish a fair price negotiation program, put in protections from excessive price increases under the Medicare program, and establish an out-of-pocket maximum for enrollees of Medicare Part D (the prescription drug program under Medicare). Under the bill, the US Health and Human Services Secretary would be required to select and negotiate the prices of at least 25 negotiation-eligible drugs per year and would be authorized to negotiate prices for up to 250 negotiation-eligible branded drugs per year by directly negotiating with manufacturers to establish a maximum price. Those branded negotiation-eligible drugs would be those that have been identified by the US federal government as costing the most to the federal government and for which there is no generic or biosimilar competition. The maximum negotiated price would be capped at 120% based on international pricing from the average price paid by Australia, Canada, France, Germany, Japan and the UK. If there is no international price, which is often the case with a new drug, under the bill the maximum fair price negotiated for a selected drug would generally be 85% of the average manufacturer price. The negotiated prices would be available to all purchasers not just Medicare beneficiaries. The proposed legislation would also impose non-compliance fees on companies that do not enter into negotiations by implementing a tax on the manufacturer’s annual gross sales beginning with 65%, plus an additional 10% for every quarter the manufacturer does not comply, to a maximum of 95%.
In an earlier analysis, the Congressional Budget Office (CBO) estimated that provisions in the bill requiring drug price negotiations and providing a source of pressure on manufacturers to secure price concessions would have reduced federal spending by $456 billion. For new drug development, CBO estimated that H.R. 3 would have reduced global revenue for new drugs by 19%, leading to approximately eight fewer drugs introduced to the US market over the 2020–2029 period (a 3% reduction) and 30 fewer drugs over the next decade (a 10% reduction).
Updated CBO analysis
The CBO recently updated its model used to inform estimates of the effects of H.R. 3 on the number and timing of new drugs entering the US market and issued a working paper, CBO’s Simulation Model of New Drug Development, to describe the update to its model. To illustrate how the model works, CBO examines a policy that reduces expected returns of drugs in the top quintile of expected returns by 15% to 25%. That policy is estimated to lead to two fewer drugs in the first decade (a reduction of 0.5%), 23 fewer over the next decade (a reduction of 5%) and 34 fewer drugs in the third decade (a reduction of 8%).
It further considers a policy that that both reduces expected returns by 15% to 25% and reduces available cash to the industry by $900 billion. CBO calculates that the reduction in cash available increases the weighted average cost of capital (WACC) by 20 basis points (from a discount rate of 8.6% to 8.8%). The combined effect is that 9% fewer new drugs will enter the market in the third decade under the policy, increasing the size of the effect by 1 percentage point.
Additional factors in future analysis
In its model, the CBO outlines various forms of uncertainty affecting estimates. In addition to the uncertainty associated with the value of the model’s parameters, inherent uncertainty is associated with the drug-development process. Uncertainty also exists in how manufacturers react to the major change in the government’s role in price determination. The simulations under the model illustrate effects by using a particular set of revenue reductions (15% to 25%) as inputs. In later analyses, CBO expects to use inputs stemming from specific legislation that are the agency’s best estimate of the firm’s expectations about average revenue reductions in the future; the firm’s own estimates may be larger or smaller than CBO’s. In the future, CBO could use the model to estimate whether and how various policies would affect the development of new drugs. CBO may adjust the model to account for how policies influence the process of new drug development.
The model also factors in a bio/pharma company’s decision-making along the various phases of clinical development, including the expected cost and expected returns of entering a given phase of development and what happens when a policy is introduced that reduces the top quintile of expected returns by 15% to 25%. Using the model, CBO estimates that such a policy would reduce the number of drugs entering the market by 0.5% in the first decade under the policy. Owing to an accumulated effect through the phases, CBO estimates the number of drugs entering the market decreases by 8% in the third decade under the policy.
Industry feedback
The CBO model provides information on the policy debate on how best to address the cost of prescription drugs while maintaining product innovation in the bio/pharmaceutical industry. To that end, in responding to the CBO report, Stephen J. Ubl, President and CEO of the Pharmaceutical Research and Manufacturers of America (PhRMA), which represents innovator, research-based bio/pharmaceutical companies, briefly outlined policy alternatives to those found in H.R. 3. “…[P]olicymakers should work with us on real solutions like capping out-of-pocket costs in Part D, lowering cost sharing and spreading those costs over the calendar year, and making sure the savings negotiated with health plans are passed to patients,” he said in an August 27, 2021 statement. “These solutions would lower costs at the pharmacy while also ensuring a future where new treatments are available for patients in need.”
Other legislative proposals
Earlier this year (March 2021), Congress introduced three bills that build on measures in H.R. 3, which passed the House of Representatives in the last Congress in December 2019. The legislative package includes three bills (see Figure 1)
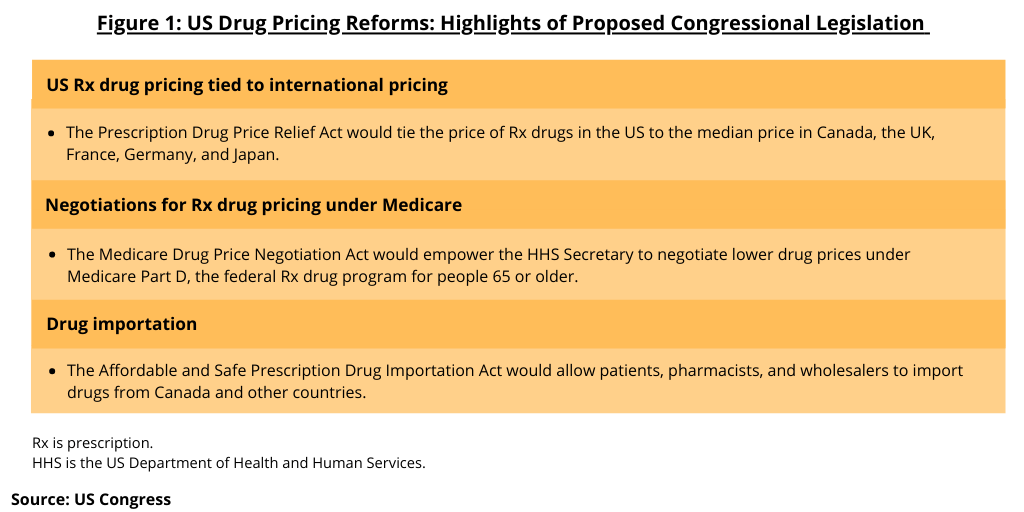
These include: (1) The Prescription Drug Price Relief Act that would tie the price of prescription drugs in the US to the median price in Canada, the UK, France, Germany, and Japan; (2) The Medicare Drug Price Negotiation Act would authorize the Secretary of the US Department of Health and Human Services (HHS) to negotiate lower drug prices under Medicare Part D, the federal prescription drug program for people 65 or older; and (3) The Affordable and Safe Prescription Drug Importation Act that would allow patients, pharmacists, and wholesalers to import drugs from Canada and other countries. The bills were introduced in the Senate by Senator Bernie Sanders (I-VT), and in the House of Representatives by Reps. Ro Khanna, (D-CA), Lloyd Doggett (D-TX), Peter Welch (D-VT), and Cori Bush (D-MO), along with more than two dozen co-sponsors.